Subscribe
The Challenge of Seasonal Influenza and Vaccine Timing
Seasonal influenza remains one of the world’s most persistent infectious threats, causing an estimated 291,000 to 646,000 respiratory deaths each year and infecting 5–15% of the global population. Despite coordinated global surveillance and biannual vaccine strain recommendations from the World Health Organization (WHO), vaccine effectiveness continues to fluctuate, in part because the virus evolves rapidly and unpredictably. Improving how and when vaccine strains are selected could substantially reduce illness and death worldwide.The challenge begins with timing. Most seasonal influenza vaccines target the immunodominant hemagglutinin (HA) surface protein and are produced in chicken eggs. Because egg-based manufacturing requires approximately six to eight months, the WHO convenes twice yearly—once in February for the northern hemisphere and once in September for the southern hemisphere—to recommend strains for the upcoming season. For each cycle, one strain is selected for each major subtype: A/H3N2, A/H1N1pdm, and B/Victoria. However, new viral variants can emerge after these selections are made, creating mismatches between vaccine strains and the viruses that ultimately circulate.
Assessing Strain Selection and Predicting Viral Evolution
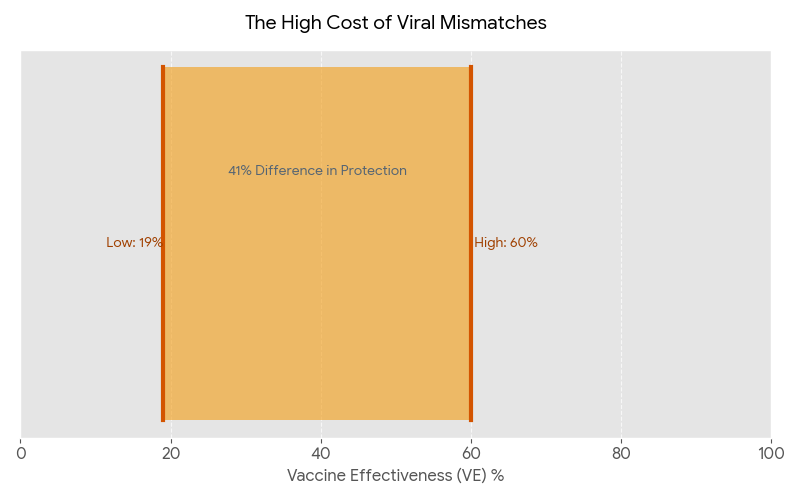
These mismatches have measurable consequences. In the United States, vaccine effectiveness (VE) estimates ranged from 19% to 60% between the 2010 and 2023 seasons (excluding the COVID-19–disrupted 2020–2021 season). And 10% – 60% from the 2004 – 2025 seasons according to the CDC. A major contributor to this variability is the rapid evolution of A/H3N2, the fastest-evolving influenza subtype and one that has dominated circulation in many seasons since its emergence in humans in 1968.
To better understand how timing affects vaccine accuracy, researchers analyzed influenza seasons from 2002 through 2023 across the United States, Europe, Australia, and New Zealand. For each season, they identified the dominant circulating viral clade—defined as the most prevalent genetic lineage—during peak transmission periods (October to April in the northern hemisphere and April to September in the southern hemisphere). For northern hemisphere strain selection in February, they incorporated global consensus A/H3N2 HA sequences collected in December and January to assess how more recent data might influence strain choice.
The study found that shortening the forecasting horizon—from 12 months to three months—reduced prediction errors in clade frequency by approximately 50%. In other words, selecting strains closer to the start of flu season substantially improved the ability to anticipate which viral lineages would dominate. Another study by different researchers concluded the same. Moreover, using a newly developed “reproducible selection method” based on a global two-month HA consensus improved vaccine match in 43 out of 63 regional seasons when evaluated by the number of classically defined epitope mutations. When applied more broadly, matching vaccines to circulating viruses based on epitope mutations could have been improved in 51 of 63 seasons while still preserving current WHO timing.
Opportunities with New Vaccine Technologies
Implementing later strain selection poses logistical barriers for traditional vaccines. Because egg-based production requires long lead times, delaying selection would be operationally difficult. Newer vaccine technologies may offer a solution. Recombinant and mRNA platforms have significantly shorter production timelines, potentially allowing strain selection to occur closer to flu season. Encouragingly, seasonal influenza mRNA vaccines have demonstrated safety profiles comparable to licensed egg-based vaccines and have elicited statistically noninferior—or in some cases superior—immune responses.
Another important insight from the study concerns update frequency. Currently, vaccine strains are revised only when expert consensus determines that an update is necessary, meaning some years pass without changes. The findings suggest that selecting new vaccine strains annually—even when changes appear modest—often results in closer genetic matches between vaccine and circulating viruses. Further gains could be achieved by modestly delaying strain selection, particularly if supported by faster manufacturing technologies.
Together, these findings underscore a central tension in influenza control: the need to balance predictive accuracy with production realities. As vaccine technology evolves, public health agencies may gain the flexibility to refine strain selection timing and methodology. Doing so could narrow the gap between vaccine composition and viral evolution—ultimately improving protection against one of the world’s most enduring seasonal threats.